Rong Wang, PhD
Biography
MOLECULAR PROGRAMMING OF THE VASCULATURE IN DEVELOPMENT AND DISEASE
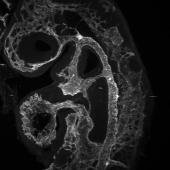
Proper formation and function of the vasculature are crucial for health and survival, as the vasculature supplies all cells in the body. A dysfunctional vasculature causes myriad diseases, including stroke, arterial occlusive diseases, and vascular anomalies. Our long-term goal is to identify novel drug targets and inform rational therapeutic designs to treat vascular diseases. Our strategy is to understand genes crucial for angiogenesis (new vessel formation) in the normal and diseased states, concentrating on the Notch, ephrin-B2, and TGF-beta pathways. We employ cutting-edge mouse genetics to delete or express genes in a cell lineage-specific and temporally controllable fashion in vascular cells. We combine these molecular approaches with mouse models of diseases as well as live 5D two-photon imaging (3D + blood flow over time) to uncover both the molecular mechanisms and hemodynamic signals in development and disease progression. These preclinical animal studies are coupled with patient sample validations. Our lab members come from diverse fields, including biology, bioengineering, and medicine, creating a collaborative and exciting environment. We strive to advance multiple projects across disciplines.
PROJECTS
Molecular programming of blood vessels: Building on our study of the developing dorsal aorta and cardinal vein, the first major artery-vein (AV) pair to form in the body, our lab aims to identify molecular regulators that program arteries and veins in vital organs during development and aging. We examine the interplay between genetic AV programming and flow-induced patterning. Understanding AV programming in normal angiogenesis provides important insights into how the genetic pathways can be hijacked in various disease states.
Stroke: We study two types of stroke, ischemic stroke and hemorrhagic stroke. Ischemic stroke occurs when arteries supplying the brain are blocked. Using a surgical model of ischemic stroke, we aim to identify technologies enabling better recover following arterial blockade. Hemorrhagic stroke, on the other hand, occurs when diseased blood vessels rupture. Brain arteriovenous malformations (AVMs), which are direct connections from arteries to veins, are one of the major causes of hemorrhagic stroke. We investigate AV programming in both AVM progression and regression.
Arteriovenous malformations: AVMs can occur anywhere in the body and comprise a category of hard to treat vascular anomalies. Most AVMs are sporadic, thus limiting the understanding of their etiology. In contrast, hereditary AVMs, such as those found in hereditary hemorrhagic telangiectasia (HHT) patients, offer an excellent opportunity to study how AVMs form. HHTs are caused by mutations in genes of the TGF-beta superfamily. We are interested in the molecular mechanisms underlying HHT-mediated AVMs formation.
Arterial occlusive diseases: Arterial occlusive diseases occur when the arteries in the body are blocked, causing insufficient blood flow to the tissues. Blockage of arteries in the brain causes stroke, in the heart causes myocardial infarction, and in the extremities causes peripheral arterial disease. Arteriogenesis, a process by which small dormant arteries around the blockage enlarge to form collateral circulation, holds promise to restore blood flow and rescue affected tissues. We investigate pro-arteriogenic molecular regulators to uncover potential therapeutic strategies to enhance the body’s natural defense against arterial occlusive disease.
Publications
- Zhang Y, Fang C, Wang RE, Wang Y, Guo H, Guo C, Zhao L, Li S, Li X, Schultz PG, Cao YJ, Wang F. A tumor-targeted immune checkpoint blocker. Proc Natl Acad Sci U S A. 2019 08 06; 116(32):15889-15894.
- Cuervo H, Nielsen CM, Simonetto DA, Ferrell L, Shah VH, Wang RA. Endothelial notch signaling is essential to prevent hepatic vascular malformations in mice. Hepatology. 2016 10; 64(4):1302-1316.
- Nielsen CM, Huang L, Murphy PA, Lawton MT, Wang RA. Mouse Models of Cerebral Arteriovenous Malformation. Stroke. 2016 Jan; 47(1):293-300.
- Murphy PA, Kim TN, Huang L, Nielsen CM, Lawton MT, Adams RH, Schaffer CB, Wang RA. Constitutively active Notch4 receptor elicits brain arteriovenous malformations through enlargement of capillary-like vessels. Proc Natl Acad Sci U S A. 2014 Dec 16; 111(50):18007-12.
- Lin Y, Jiang W, Ng J, Jina A, Wang RA. Endothelial ephrin-B2 is essential for arterial vasodilation in mice. Microcirculation. 2014 Oct; 21(7):578-86.
- Nielsen CM, Cuervo H, Ding VW, Kong Y, Huang EJ, Wang RA. Deletion of Rbpj from postnatal endothelium leads to abnormal arteriovenous shunting in mice. Development. 2014 Oct; 141(19):3782-92.
- Lindskog H, Kim YH, Jelin EB, Kong Y, Guevara-Gallardo S, Kim TN, Wang RA. Molecular identification of venous progenitors in the dorsal aorta reveals an aortic origin for the cardinal vein in mammals. Development. 2014 Mar; 141(5):1120-8.
- Costa MJ, Wu X, Cuervo H, Srinivasan R, Bechis SK, Cheang E, Marjanovic O, Gridley T, Cvetic CA, Wang RA. Notch4 is required for tumor onset and perfusion. Vasc Cell. 2013 Apr 20; 5(1):7.
- Kim TN, Goodwill PW, Chen Y, Conolly SM, Schaffer CB, Liepmann D, Wang RA. Line-scanning particle image velocimetry: an optical approach for quantifying a wide range of blood flow speeds in live animals. PLoS One. 2012; 7(6):e38590.
- Murphy PA, Kim TN, Lu G, Bollen AW, Schaffer CB, Wang RA. Notch4 normalization reduces blood vessel size in arteriovenous malformations. Sci Transl Med. 2012 Jan 18; 4(117):117ra8.
- Miniati D, Jelin EB, Ng J, Wu J, Carlson TR, Wu X, Looney MR, Wang RA. Constitutively active endothelial Notch4 causes lung arteriovenous shunts in mice. . 2010 Feb; 298(2):L169-77.
- Herbert SP, Huisken J, Kim TN, Feldman ME, Houseman BT, Wang RA, Shokat KM, Stainier DY. Arterial-venous segregation by selective cell sprouting: an alternative mode of blood vessel formation. Science. 2009 Oct 09; 326(5950):294-8.
- Murphy PA, Lu G, Shiah S, Bollen AW, Wang RA. Endothelial Notch signaling is upregulated in human brain arteriovenous malformations and a mouse model of the disease. Lab Invest. 2009 Sep; 89(9):971-82.
- Yang Y, Tang G, Yan J, Park B, Hoffman A, Tie G, Wang R, Messina LM. Cellular and molecular mechanism regulating blood flow recovery in acute versus gradual femoral artery occlusion are distinct in the mouse. J Vasc Surg. 2008 Dec; 48(6):1546-58.
- Kim YH, Hu H, Guevara-Gallardo S, Lam MT, Fong SY, Wang RA. Artery and vein size is balanced by Notch and ephrin B2/EphB4 during angiogenesis. Development. 2008 Nov; 135(22):3755-64.
- Murphy PA, Lam MT, Wu X, Kim TN, Vartanian SM, Bollen AW, Carlson TR, Wang RA. Endothelial Notch4 signaling induces hallmarks of brain arteriovenous malformations in mice. Proc Natl Acad Sci U S A. 2008 Aug 05; 105(31):10901-6.
- Dubois NC, Adolphe C, Ehninger A, Wang RA, Robertson EJ, Trumpp A. Placental rescue reveals a sole requirement for c-Myc in embryonic erythroblast survival and hematopoietic stem cell function. Development. 2008 Aug; 135(14):2455-65.
- He C, Hu H, Braren R, Fong SY, Trumpp A, Carlson TR, Wang RA. c-myc in the hematopoietic lineage is crucial for its angiogenic function in the mouse embryo. Development. 2008 Aug; 135(14):2467-77.
- Carlson TR, Hu H, Braren R, Kim YH, Wang RA. Cell-autonomous requirement for beta1 integrin in endothelial cell adhesion, migration and survival during angiogenesis in mice. Development. 2008 Jun; 135(12):2193-202.
- Tward AD, Jones KD, Yant S, Cheung ST, Fan ST, Chen X, Kay MA, Wang R, Bishop JM. Distinct pathways of genomic progression to benign and malignant tumors of the liver. Proc Natl Acad Sci U S A. 2007 Sep 11; 104(37):14771-6.
- Braren R, Hu H, Kim YH, Beggs HE, Reichardt LF, Wang R. Endothelial FAK is essential for vascular network stability, cell survival, and lamellipodial formation. J Cell Biol. 2006 Jan 02; 172(1):151-62.
- Yan J, Tang GL, Wang R, Messina LM. Optimization of adenovirus-mediated endothelial nitric oxide synthase delivery in rat hindlimb ischemia. Gene Ther. 2005 Nov; 12(22):1640-50.
- Proctor JM, Zang K, Wang D, Wang R, Reichardt LF. Vascular development of the brain requires beta8 integrin expression in the neuroepithelium. J Neurosci. 2005 Oct 26; 25(43):9940-8.
- Carlson TR, Yan Y, Wu X, Lam MT, Tang GL, Beverly LJ, Messina LM, Capobianco AJ, Werb Z, Wang R. Endothelial expression of constitutively active Notch4 elicits reversible arteriovenous malformations in adult mice. Proc Natl Acad Sci U S A. 2005 Jul 12; 102(28):9884-9.
- Carpenter B, Lin Y, Stoll S, Raffai RL, McCuskey R, Wang R. VEGF is crucial for the hepatic vascular development required for lipoprotein uptake. Development. 2005 Jul; 132(14):3293-303.
- Tang GL, Chang DS, Sarkar R, Wang R, Messina LM. The effect of gradual or acute arterial occlusion on skeletal muscle blood flow, arteriogenesis, and inflammation in rat hindlimb ischemia. J Vasc Surg. 2005 Feb; 41(2):312-20.
- Tward AD, Jones KD, Yant S, Kay MA, Wang R, Bishop JM. Genomic progression in mouse models for liver tumors. Cold Spring Harb Symp Quant Biol. 2005; 70:217-24.
- Tang G, Charo DN, Wang R, Charo IF, Messina L. CCR2-/- knockout mice revascularize normally in response to severe hindlimb ischemia. J Vasc Surg. 2004 Oct; 40(4):786-95.
- Chang DS, Su H, Tang GL, Brevetti LS, Sarkar R, Wang R, Kan YW, Messina LM. Adeno-associated viral vector-mediated gene transfer of VEGF normalizes skeletal muscle oxygen tension and induces arteriogenesis in ischemic rat hindlimb. Mol Ther. 2003 Jan; 7(1):44-51.
- Wang R, Ferrell LD, Faouzi S, Maher JJ, Bishop JM. Activation of the Met receptor by cell attachment induces and sustains hepatocellular carcinomas in transgenic mice. J Cell Biol. 2001 May 28; 153(5):1023-34.
- Wang R, Kobayashi R, Bishop JM. Cellular adherence elicits ligand-independent activation of the Met cell-surface receptor. Proc Natl Acad Sci U S A. 1996 Aug 06; 93(16):8425-30.
- Wang R, Siegal GP, Scott DL, Bautch VL. Developmental analysis of bone tumors in polyomavirus transgenic mice. Lab Invest. 1994 Jan; 70(1):86-94.
- Wang R, Clark R, Bautch VL. Embryonic stem cell-derived cystic embryoid bodies form vascular channels: an in vitro model of blood vessel development. Development. 1992 Feb; 114(2):303-16.
- Dubois NA, Kolpack LC, Wang R, Azizkhan RG, Bautch VL. Isolation and characterization of an established endothelial cell line from transgenic mouse hemangiomas. Exp Cell Res. 1991 Oct; 196(2):302-13.
- Wang R, Bautch VL. The polyomavirus early region gene in transgenic mice causes vascular and bone tumors. J Virol. 1991 Oct; 65(10):5174-83.